Software
For my research I rely greatly on a wide range of software and image analysis tools developed by computer specialists. Using these greatly increase throughput, accuracy and reproducibility in plant phenotyping. Below I provide links to all of the software I commonly use and how I use them. Before embarking on a project I strongly recommend checking out quantitative-plant.org as it is a central resource maintained by @guillaumelobet for available software and datasets.
RhizoVision Explorer Root image analysis software for quantification of disconnected or connected root systems.
FREE DOWNLOAD
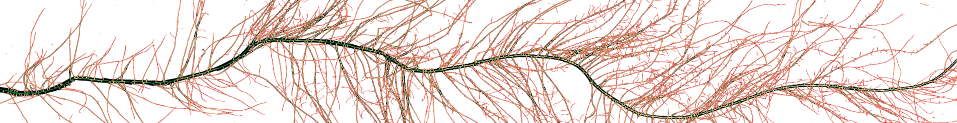
ORS Dragonfly Image analysis software with deep learning for analysing data collected from X-ray CT.
Academic/Non-profit license
RootPainter Image analysis software with deep learning for analysing Rhizotron images.
FREE DOWNLOAD

FieldImageR Image analysis scripts for analysing drone images.
FREE DOWNLOAD

PRIMAL Machine learning pipeline (Random Forest) bundled into a R Shiny app for root system architecture.
FREE DOWNLOAD

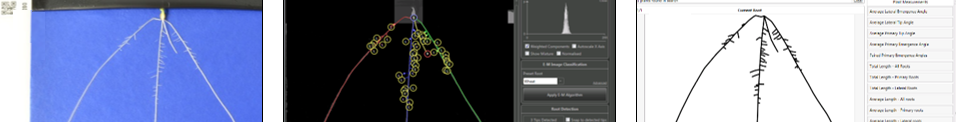
RootNav Semi-automatic software for quantification of 2D root system architecture. I have used RootNav for processing multiple mapping populations grown on hydroponic pouches. This approach is particularly useful for root angle determination. Also check out RootNav 2.0 deep learning version.
FREE DOWNLOAD
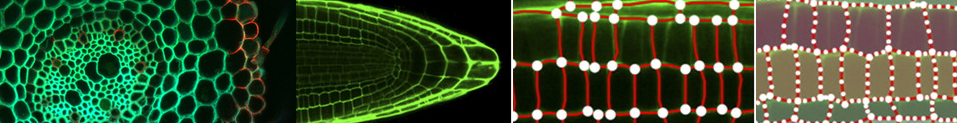
CellSeT Semi-automatic software for quantifying root anatomy by segmenting out plant cells and assigning cell classes.
FREE DOWNLOAD
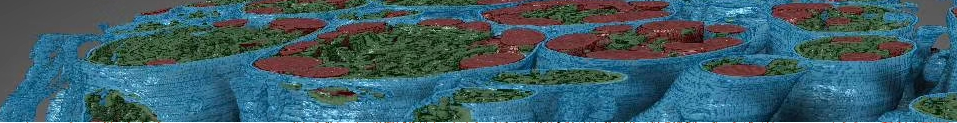
RooTrak 3D Root segmentation software for X-ray Micro Computed Tomography (µCT) scanned plants.
FREE DOWNLOAD

RooTh 3D Root skeletonisation and root system quantification software for X-ray Micro Computed Tomography (µCT) scanned plants.
FREE DOWNLOAD

OpenSimRoot 3D Root simulation software that can integrate X-ray Micro Computed Tomography (µCT) & MRI scanned plants.
FREE DOWNLOAD

Fiji is just imageJ Fiji is an image processing package, basically imageJ bundled with a lot of plugins which facilitate scientific image analysis. I personally use it to batch transform images (eg. crop, colour threshold) using the macro feature. There are many applications including measuring, skeletonisation and adding scale bars to images.
FREE DOWNLOAD
![]()
OpenCV/PlantCV Fiji may be a good starting point for image analysis but I have since transitioned to OpenCV and PlantCV functions with Jupyter notebooks.
FREE DOWNLOAD
![]()
RStudio Statistics program for data processing and graphing. RStudio makes R easier to use. Notable packages inlcude "tidyverse" and "qtl". See My Code for my R code repository.
FREE DOWNLOAD

Lab Mate - Molarity & Dilution This is a simple molarity and dilution universal app that I made for iPhone, iPad and macOS. Feel free to contact me for a code.
